Table of Contents
ES Systems’ thermal gas mass flow meters measure flow in ln/min. This unit looks like a volumetric flow unit, but in truth expresses mass flow. In this article we will explain why.
Defining volume & mass flow
Wondering what is mass flow and what is volumetric flow? Let’s see it this way:
Assume you have a 1.0 litre cylinder with a movable piston of negligible weight that closes it. Secondly, assume that this cylinder holds 1.0 litre of air at ambient pressure (1 atm) and 0oC temperature. This volume of air weights 1.293g, which equals the mass of the air enclosed to the cylinder.
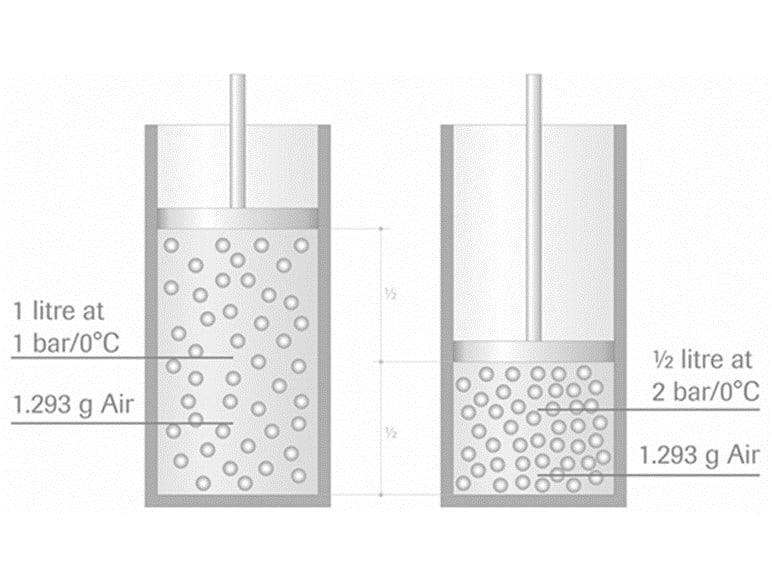 When we move the piston halfway to the bottom of the cylinder, the volume of air contained is only 1/2 liters, the pressure is increased to around 2 bar, but the mass remains the same, 1.293g, as nothing has been added or removed from the cylinder.
When we move the piston halfway to the bottom of the cylinder, the volume of air contained is only 1/2 liters, the pressure is increased to around 2 bar, but the mass remains the same, 1.293g, as nothing has been added or removed from the cylinder.
Mass & volumetric flow standards
Following the example above, you would consider that mass flow should be stated in weight units like g/h, mg/s, and so on. The majority of users, on the other hand, think and operate in volume terms. Therefore, in order to maintain this principle, you can express mass flow in volume terms as long as the conditions for converting mass to volume are agreed upon (in other words if you agree on the “reference conditions”).
According to the ‘European’ standard, a temperature of 0°C and a pressure of 1,013 bar is defined as “normal” reference conditions, indicated by the underlying letter “n” in the unit of volume used (ln/min, m3n/h, etc).
A temperature of 20°C and a pressure of 1,013 bar are used as “standard” reference conditions, indicated by the underlying letter “s” in the unit of volume used (ls/min, m3s/h).
For the ‘American’ standard, the prefix “s” in sccm, slm or scfh refers to “standard” conditions 101.325 kPa absolute (14.6959 psia) and temperature of 0°C (32°F).
Please note: Real caution should be taken in the measurement units each sensor is calibrated because, if the difference is not considered, it may result to an error of 7% – considering the high levels of precision and low total error band required in flow sensors for medical applications, for example, one can understand how important this is!
Further reading: How to calculate gas flow rate from a pressure measurement?
Differences between volumetric and mass flow meters
Variable area meters and turbine flow meters are volumetric measurement devices that are prone to temperature or pressure variations. Additional mass flow sensors for these parameters, as well as a flow computer to correct for fluctuations in these process conditions, would be required for mass flow measurement. On the other hand, temperature and pressure fluctuations have little effect on thermal mass flow meters.
Mass flow technology compensates for absolute measurements, and therefore it has an inherent advantage over volumetric flow technology. The “Ideal Gas Law” applies to common process gases such as air, argon, CO2, N2, and oxygen. While not all gases are “ideal,” most typical gases operate on the basis that temperature and pressure induce changes in density. Due to the fact that changes in temperature and pressure alter the gas density and thus diminish the accuracy, volumetric air flow is less reliable than mass flow in accounting for absolute measurements. To estimate the true gas mass flow rate, volumetric flow measurement requires temperature and pressure adjustment.
Further reading: Gas Flow Measurement – Different Types of Flow Meters
Calculating volumetric flow rate to mass flow rate
A volumetric flow at standard conditions translates to a specific mass flow rate. For example, 800 cm3/min of air at standard conditions of temperature and pressure calculates to a mass flow of 1.033 g/min. This can be calculated as follows:
Definitions:
P = Pressure [hPa][atm]
V = Volume [cm3]
n = Number of molecules of gas [mole]
R = Universal gas constant [(m3 x Pa) / (mole x K)]
T = Absolute temperature [K]
ρ = Gas density [g/cm3]
m = Mass [g]
m = Mass flow [g/min]
V = Volumetric flow [cm3/min]
VS = Volumetric flow at standard conditions [cm3/min]
The ideal gas law,
PV = nRT (1)
can be solved for the gas volume to get:
V = nRT / P (2)
Gas density is defined as:
ρ = m / V (3)
Substituting equation (1) into equation (2) redefines gas density as:
ρ = mP / nRT
Mass flow is equal to density times volumetric flow rate:
ṁ = ρ V̇ (4)
With equation (3) mass flow can be redefined as:
ṁ = mP V̇ /nRT (5)
For a volumetric flow rate of VS = 800 cm3/min at standard conditions of 273.15 K and 1 atm the true mass flow then calculates to:
ṁ = 1.033 g/min
where,
VS = 200 cm3/min
m = 28.949 grams in 1 mole air
n = 1 Mol
P = 1 atm (1013.25 hPa)
R = 82.1 (cm3 x atm) / (mole x K)
T = 273.15 K (0 °C)
Calculating mass flow rate to volumetric flow rate
By rearranging equation (5) the corresponding volumetric flow at nonstandard conditions of 25 °C can be calculated for the mass flow as follows:
V̇ = ṁ nRT / mP (6)
Considering the same example as above we see that:
V̇ = 873.5 cm3/min
where,
ṁ = 1.033 g/min
m = 28.949 grams in 1 mole air
n = 1 Mol
P = 1 atm (1013.25 hPa)
R = 82.1 (cm3 x atm) / (mole x K)
T = 298.15 K (25 °C)
As it can be deduced from above, a change of 25°C in temperature will result in 73.5 cm3/min volumetric flow difference in measurement while the mass flow would remain the same. This can be explained as with higher temperature, the gas expands. Therefore, a higher volumetric flow rate is required to pass the same number of molecules (mass).
Check out ES Systems’ free online Pressure Units Converter for easier conversions on the go.



